Manufacturing and Mass Production of Metal Nanoparticles
New Trends in Metal Nanoparticle Manufacturing
(Patent No. 4688796)
Nanoparticles have extremely large specific surface areas and may exhibit properties that differ from bulk solid materials, and research and application development is progressing in various fields. In particular, focusing on metal nanoparticles, in recent years, nanoparticles of metals with low electrical resistance such as gold and copper have been used as inks for forming fine wiring on IC substrates, and platinum nanoparticles are beginning to be applied in cosmetics and food products.
Conventional methods for producing metal nanoparticles include pulverizing metal lumps with mills, methods that vaporize or sputter metals by heating them in inert gas or vacuum, and methods that reduce metal salts or thermally decompose metal complexes in liquid phases at room temperature—many manufacturing processes have been developed. On the other hand, these methods have numerous issues: the manufacturing method is extremely limited depending on the type of metal element in the metal nanoparticle, there are limits to particle size refinement and uniformity, raw materials and equipment are costly, productivity is poor, and so on.
I’MSEP is developing technology to produce nanoparticles of any metal (including elements not strictly classified as "metals") with the same equipment configuration, using a new method called plasma-induced cathode electrolysis via MSEP. Plasma-induced electrolysis using discharge phenomena in molten salt systems is broadly classified into plasma-induced cathode electrolysis and plasma-induced anode electrolysis depending on the direction of current flow, but plasma-induced cathode electrolysis is mainly used when producing metal and alloy nanoparticles.
In this method, unlike conventional electrolysis, the cathode is positioned above the molten salt bath surface as shown in the following figure. When electrolysis is started, discharge occurs between the cathode and the molten salt bath surface, and electrons are supplied into the molten salt. By supplying ions of metal M, for which particle formation in the molten salt is desired, the metal ions (Mn+) are reduced by discharge electrons from the cathode, and metal nanoparticles M are generated in the molten salt near the bath surface.
Cathode: Mn+ + n e- → M
Because the electrolysis operation is as shown in the figure, it can be implemented with a very simple equipment configuration, and complex vacuum equipment is not required. In addition, the voltage between the cathode and the molten salt bath surface is on the order of several tens of volts under steady-state conditions, and it is not necessary to apply high voltage at all times.
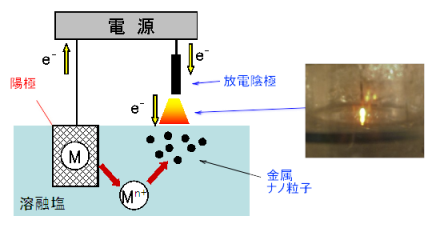 Figure: Principle of plasma-induced cathode electrolysis (example)
Figure: Principle of plasma-induced cathode electrolysis (example)
Manufacturing Nanoparticles of Any Metal
Using this method, which forms particles through bottom-up from the atomic level, it is possible to control the entire process from atom generation through nucleation to the start of particle growth. It is precisely because molten salt can handle electrochemical reactions of any element that nanoparticles can be formed with the same equipment configuration—and extremely fine ones at that—even for elements that are difficult with existing methods, such as high-melting-point, high-hardness metals like Ti, Ta, and W, or non-metals such as Si, B, and C. In addition, since the formation rate of metal nanoparticles is proportional to the current passed, control of the formation rate is also easy.
Recycling and Value-Adding of Scarce Metals Such as Tantalum
As shown in the principle diagram above, by using a metal anode containing metal M, for example, metal ions (Mn+) can be supplied into the electrolytic bath through electrochemical anodic dissolution.
Anode: M → Mn+ + n e-
That is, the bulk metal used for the anode becomes the raw material for metal nanoparticles. Since scrap metal and other waste materials can be used for this anode material, recycling and value-adding of scarce metals can be achieved simultaneously.
For example, in the case of scarce metals such as tantalum, where resource depletion and geographic concentration are concerns, tantalum scrap from manufacturing can be directly regenerated into tantalum nanoparticles. Such tantalum nanoparticles are expected to see growing demand as electrode materials for high-performance tantalum capacitors. We believe that the practical application of plasma-induced cathode electrolysis can greatly contribute to the continued development of the tantalum capacitor market by meeting future demands for further particle refinement, and through supply stabilization and cost reduction of nanoparticle supply.
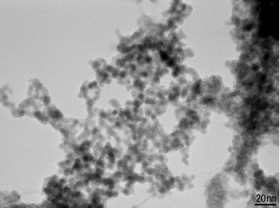 Figure: TEM image of tantalum nanoparticles formed by plasma-induced cathode electrolysis.
Figure: TEM image of tantalum nanoparticles formed by plasma-induced cathode electrolysis.
Continuous Manufacturing Process for Metal Nanoparticles
Rotating Disk as the Key to Refinement and Continuous Production
(Patent No. 4755567)
We are developing a continuous electrolysis method using a rotating disk to further refine and homogenize nanoparticles formed by plasma-induced cathode electrolysis, and to enable continuous manufacturing. By performing electrolysis on a rapidly rotating disk, it is possible to quickly recover extremely fine nanoparticles in the early growth stage and disperse and hold them in solidified salt.
We have been conducting production using small-scale test equipment, and have succeeded, for example, in forming extremely fine nickel nanoparticles with primary particle diameters of approximately 10 nm, which are expected as next-generation capacitor materials.
This new electrolysis method using a rotating disk has excellent features for practical equipment development: simplified equipment configuration, improved continuous productivity and workability, recovery of metal nanoparticles in dispersed form, reduced molten salt usage, and ease of scale-up. Currently, it has been selected for the JST Research Results Optimal Deployment Support Program (A-STEP) SME/Venture Development Theme, and equipment development toward early practical application is proceeding at a rapid pace.
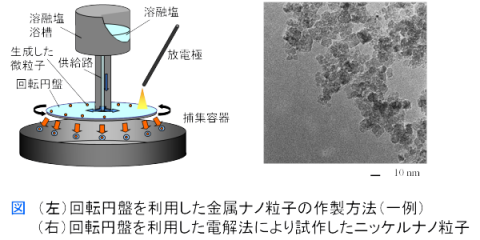
 Figure: Nanoparticle continuous manufacturing equipment currently under development (JST A-STEP selected project)
Figure: Nanoparticle continuous manufacturing equipment currently under development (JST A-STEP selected project)
Direct Production of Si Nanoparticles from SiO2 Powder
Nanoparticle Generation Method Using Metal Oxide Powder as Raw Material
(PCT/JP2011-73877)
The application range of plasma-induced cathode electrolysis is expanding further, and it has become possible to directly form metal nanoparticles using metal oxide powder as raw material, which is relatively inexpensive and readily available. This eliminates the need to prepare high-priced bulk metal or metal halides that dissolve easily in molten salt as raw materials.
For example, by suspending SiO2 powder in molten salt and performing plasma-induced cathode electrolysis, the SiO2 powder in the molten salt is electrochemically reduced directly, and Si nanoparticles are formed.
Cathode: SiO2 + 4 e- → Si + 2 O2-
Such Si nanoparticles are expected to find applications in lithium-ion battery anodes, solar cells, and elsewhere.
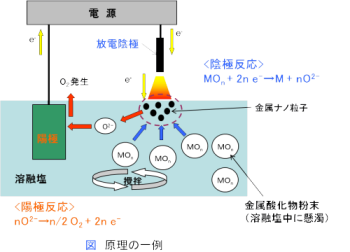
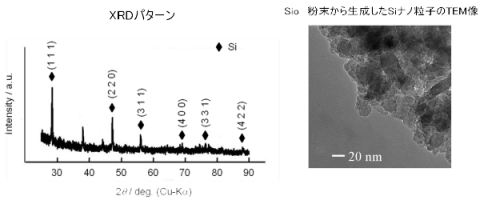
Figure: TEM image and XRD pattern of Si nanoparticles generated from silicon dioxide powder
Creation of Various Functional Nanoparticles
Formation of Alloy Particles Using Displacement Reactions in Molten Salt
(Patent No. 5065948)
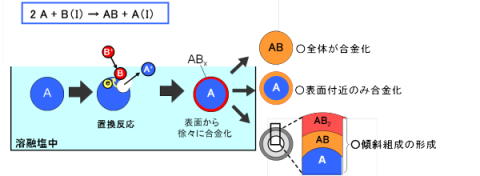
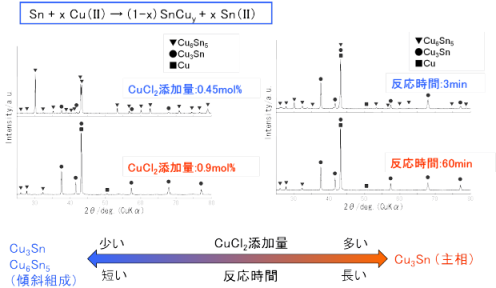
As described in another section, plasma-induced cathode electrolysis can directly form multicomponent alloy nanoparticles by adding multiple types of metal ions to the molten salt and reducing them simultaneously. On the other hand, alloy particles can also be formed using displacement reactions in molten salt. For example, Sn-Cu alloy nanoparticles can be formed by reacting Sn nanoparticles with Cu ions in molten salt.
Such Sn-based and Si-based alloy nanoparticles are expected as anode materials for lithium-ion secondary batteries. In addition, we have succeeded in forming various alloy nanoparticles including FePt nanoparticles with excellent magnetic properties.
Since composition control is easy and alloy particle manufacturing is possible in a simple and low-cost manner, this technology has high potential as a new technology surpassing conventional mechanical alloying methods, and early practical application is expected.
When Forming Alloy Particles of Metal A and Metal B (B is Nobler than A)
Formation of Metal Compound Particles by Plasma-Induced Anode Electrolysis
In plasma-induced anode electrolysis, the anode is positioned above the electrolytic bath surface, and various compound particles can be formed by performing anode electrolysis with molten salt containing the anion of the raw material. For example, by using titanium as the anode and performing electrolysis on molten salt containing oxide ions (O2-), titanium oxide nanoparticles can be formed.
(Example) Anode: Ti + n O2- → TiOn + 2n e-
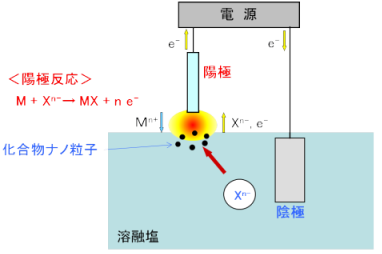 Figure: Principle of plasma-induced anode electrolysis (example);
(Reaction mechanism differs depending on the target compound)
Figure: Principle of plasma-induced anode electrolysis (example);
(Reaction mechanism differs depending on the target compound)

Rare Metal / Rare Earth Recycling
Taking Rare Earth Recycling from Nd Magnets as an Example
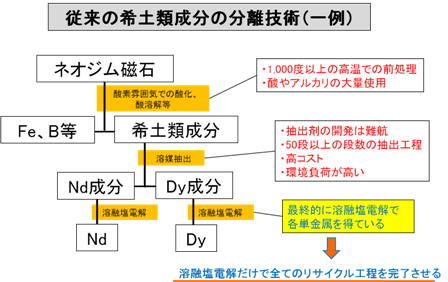
Rare earth metals have become indispensable elements in advanced industries. For example, high-performance rare earth sintered magnets (neodymium magnets) of the neodymium-iron-boron type are widely used in hybrid vehicles, air conditioners, hard disks, and other applications. On the other hand, rare earth metals have high geographic concentration, and confirmed reserves are very small for some elements, so in order for Japan to ensure a stable supply of rare earth elements for the future, it is necessary to efficiently recycle rare earth metals from scrap existing domestically.
Conventionally, in recovering rare earth components from neodymium magnet scrap, for example, the entire amount is dissolved using large quantities of acid, then iron is precipitated with alkali, and the rare earth components are recovered as oxides from the remaining solution. Furthermore, to separate and recover rare earth components by element, an extraction-separation process with 50 or more stages using expensive solvents with high environmental impact is required, which significantly affects recycling costs.
The rare earth components extracted by such solvent extraction methods are ultimately converted to rare earth oxides and the like, and even today, rare earth metals are regenerated from them by molten salt electrolysis. Molten salt electrolysis inherently excels in handling rare earth elements, as the rare earth metals such as neodymium and dysprosium that are the metal raw materials for neodymium magnets are produced by this method. Therefore, if this molten salt electrolysis must be performed as the final stage of the recycling process anyway, we consider it most preferable to complete the entire recycling process including the above-mentioned rare earth element separation with this molten salt electrolysis alone.
Electrochemical Implantation / Displantation
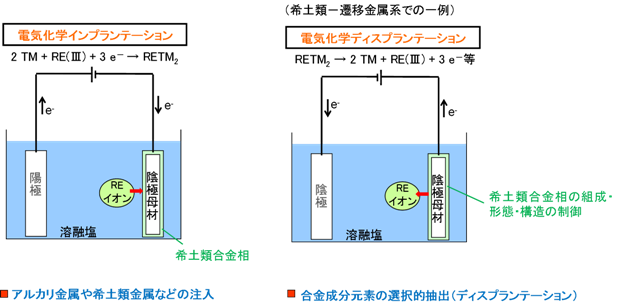
I’MSEP Co., Ltd. has accumulated extensive research data on the electrochemical behavior of rare earth elements in molten salt. We have also accumulated numerous findings on electrochemical alloying / dealloying (electrochemical implantation / displantation) of rare earth–transition metal systems.
Electrochemical Implantation
For example, it is possible to form an alloy between a substrate used as the cathode and rare earth metal by utilizing the electrochemical reduction reaction of rare earth ions in molten salt (electrochemical implantation).
Dy(III) + 2 Ni + 3 e- → DyNi2 (Example in Dy-Ni system)
Electrochemical Displantation
Furthermore, by using this rare earth alloy as an anode and electrochemically dissolving it anodically in molten salt, it is possible to selectively dissolve rare earth metals from the alloy phase as ions (electrochemical displantation). In addition, by controlling the anode potential during this process, the composition after displantation can be freely controlled from rare earth-rich alloy phases to complete dissolution to restore the original substrate composition.
3 DyNi2 → Dy(III) + 2 DyNi3 + 3 e- (Example in Dy-Ni system)
By utilizing this electrochemical implantation / displantation, it may be possible to separate Nd and Dy more efficiently (with fewer electrolysis treatment cycles) not merely by depositing and dissolving them as pure metals, but by utilizing alloy formation with an appropriate substrate metal and selective dissolution from that alloy phase.
New Recycling Technology Using Bifunctional Electrodes
(JP 2009-235552)
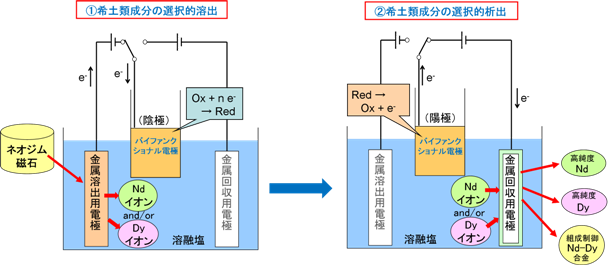
Using I’MSEP's expertise and know-how, it is possible to construct a selective recovery process for rare earth metals from neodymium magnets using molten salt electrolysis alone.
An example of the principle of this process is shown in the figure below. The electrolysis device consists of a metal dissolution electrode, a metal recovery electrode, and a bifunctional electrode.
The bifunctional electrode refers to a dual-function electrode that can serve as both a cathode and an anode as needed.
First, the neodymium magnet to be recycled is set up as the anode. Current is passed between it and the bifunctional electrode (as cathode), and by controlling the anode potential during this process, neodymium and dysprosium are selectively dissolved anodically into the molten salt while non-rare-earth magnet components (Fe, B, etc.) are left in the anode.
After the current is stopped, the bifunctional electrode is then used as the anode and the metal recovery electrode as the cathode for electrolysis. By appropriately selecting the metal used for the cathode and controlling the cathode potential, the dissolved rare earth metal ions can be selectively reduced and alloyed with the substrate metal.
The separation efficiency for each rare earth element can be improved by repeating the selective anodic dissolution and selective cathodic deposition (alloying) steps as needed, using the alloy formed at the cathode again as the anode.
In this way, it is possible to separate and recover rare earth metals such as neodymium and dysprosium contained in neodymium magnets by element, or to recover them as Nd-Dy alloy with precisely controlled composition.
Applicable to Recycling Processes for Any Scarce Element
Similarly, attractive proposals can be made for the recovery and value-adding of scarce elements from capacitor materials containing tantalum and niobium, catalyst materials containing large amounts of precious metals, semiconductor materials used in solar cell elements and LCD panels, and all other materials.
Electrolytic Nitriding Using Molten Salt
Electrolytic Nitriding Method
(JP 2009-52104, Patent No. 4471728)
At I’MSEP, using MSEP, we can create various functional surfaces by electrolysis—surface modifications such as nitriding, boriding, and carburizing of material surfaces, and coatings with metal nitrides, boron, and the like. As an example of such surface functionalization, the figure below shows a surface nitriding method by electrolysis. The conductive substrate to be surface-treated is placed in the electrolytic bath as the anode.
By electrochemically oxidizing nitride ions (N3-) added in advance to the molten salt at this anode surface, highly active nitrogen atoms are generated, which react with the metal of the substrate surface to form a nitride layer. In addition to this "N3-" reaction, there are several other reactions that can be used for nitriding treatment, so the optimal treatment method can be selected according to the type and characteristics of the material.
Furthermore, since this electrochemical nitriding reaction proceeds at all points on the substrate surface that contact the molten salt, nitriding of substrate surfaces with fine and complex shapes—such as the interior of the small cylinder shown in the figure below—is also possible.
Creation of New Materials by Low-Temperature Nitriding
By selecting the type of molten salt used, nitriding treatment at 300°C or below—which was previously difficult to achieve rapidly—becomes possible. For example, in the case of stainless steel, a stable oxide film exists on the surface, so corrosion resistance is high, but the electrical contact resistance of the surface is large.
When nitriding stainless steel at high temperature, effects such as increased surface hardness and reduced contact resistance can be obtained, but the inherent high corrosion resistance of stainless steel is lost.
However, by performing nitriding at low temperature, it is possible to reduce surface contact resistance and improve surface hardness without impairing the corrosion resistance of stainless steel. This low-temperature nitrided stainless steel is expected to find wide applications, including metal separators for fuel cells.
In this way, low-temperature nitriding may enable functional surfaces that cannot be obtained with high-temperature nitriding. Such low-temperature nitriding was a difficult treatment that could previously only be performed by limited methods, but it has become possible to carry out simply through the electrolytic nitriding method using MSEP.
Figure: Cross-sectional SEM image of SUS304 after nitriding at approximately 300°C
Figure: Electrochemical corrosion resistance test results of low-temperature nitrided specimen
(Linear sweep voltammogram, 20 mV/min, 5 wt% sulfuric acid solution, room temperature)
 Figure: Principle of plasma-induced cathode electrolysis (example)
Figure: Principle of plasma-induced cathode electrolysis (example)
 Figure: TEM image of tantalum nanoparticles formed by plasma-induced cathode electrolysis.
Figure: TEM image of tantalum nanoparticles formed by plasma-induced cathode electrolysis.

 Figure: Nanoparticle continuous manufacturing equipment currently under development (JST A-STEP selected project)
Figure: Nanoparticle continuous manufacturing equipment currently under development (JST A-STEP selected project)





 Figure: Principle of plasma-induced anode electrolysis (example);
Figure: Principle of plasma-induced anode electrolysis (example);







