RESEARCH CONTENTSResearch
I’MSEP's unique research based on
molten salt electrolysis technology
RESEARCH CONTENTSCO2 Resource Utilization

Under the goal of "achieving carbon neutrality by 2050 and realizing a decarbonized society," there is a growing social need to not merely reduce CO2 emissions as much as possible and capture emitted CO2 to minimize environmental impact through storage and management—a passive perspective—but to actively seek ways to utilize it as a resource. I’MSEP Co., Ltd. is advancing the development and social implementation of innovative technologies that make this possible, aiming for the realization of a "carbon-driven society." The fundamental reaction underlying this technology is as follows.
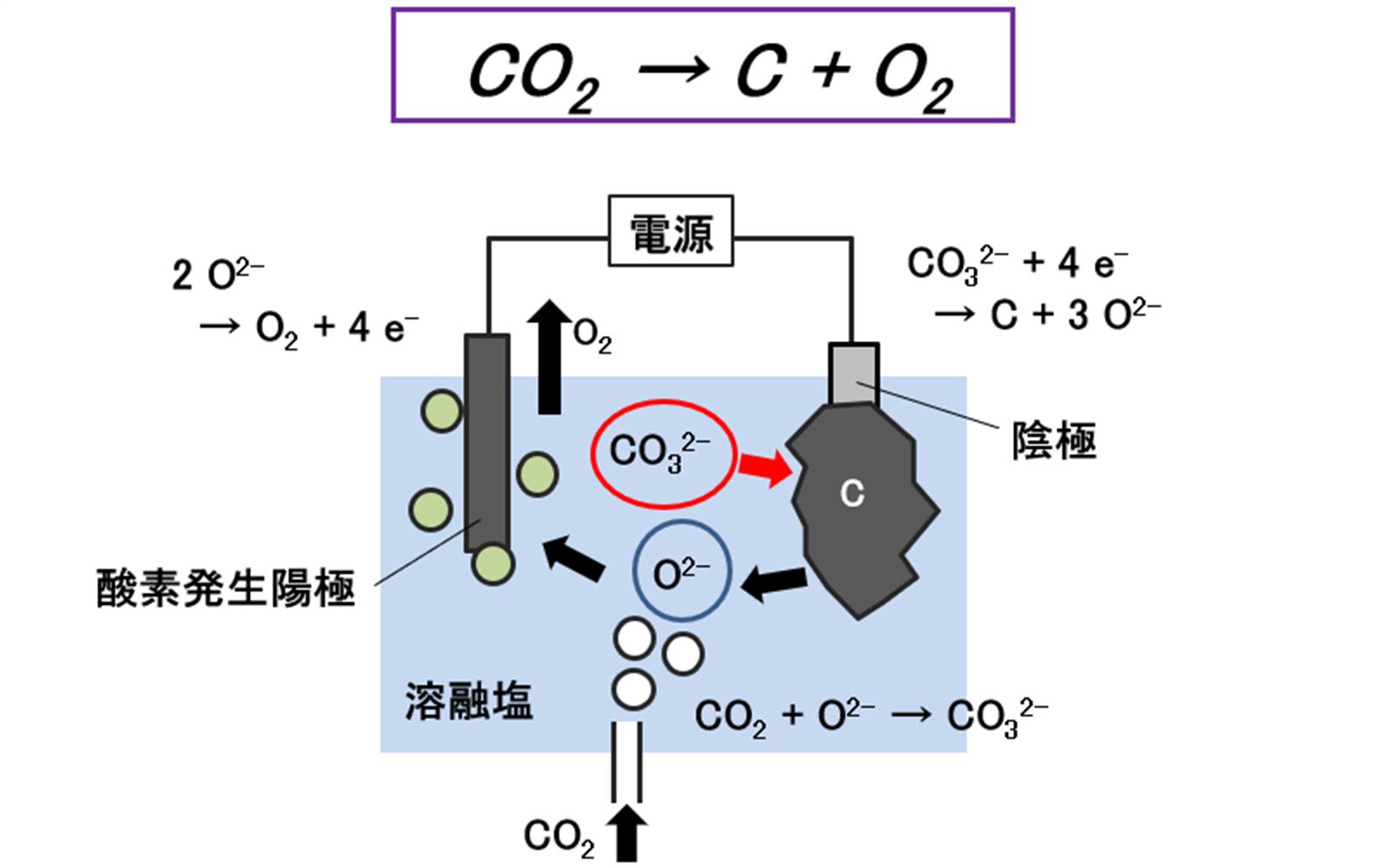
When electrolysis is performed by dissolving potassium carbonate in molten chloride, a carbon film is formed on the cathode surface.
Cathode reaction: CO32– + 4e– → C + 3 O2–
At this time, when carbon dioxide is blown into the electrolytic bath, a portion of the O2- ions reacts as follows:
CO2(g) + O2– → CO32–
and becomes CO32–, which is again reduced to carbon at the cathode. The excess O2- ions, other than those required for this reaction, become oxygen gas through the anodic reaction:
Anode reaction: 2O2– → O2↑+ 4e–
and are recovered at the anode.
Ultimately, the overall reaction is as follows:
Overall reaction: CO2 → C + O2↑
The carbon produced at the cathode can be obtained in various shapes, forms, and structures by adjusting the electrolysis conditions. Experiments have confirmed the formation of smooth, highly adherent carbon plating films.
If this system is realized, for example, CO2 emitted from thermal power plants and factories can be absorbed into molten salt and converted to carbonate ions, and electrolysis in molten salt using renewable energy can convert it into high-performance carbon materials or fix it as solid carbon fuel. Thereafter, the obtained carbon can be effectively utilized as materials for electrochemical energy conversion devices or as fuel for fuel cells (direct carbon fuel cells or integrated CO2 electrolysis–fuel cell devices). It can also be used as a reducing agent in metal refining.
I’MSEP Co., Ltd. positions this technology as an important technology that can contribute to the realization of a "CO2 resource utilization and recycling system," and is advancing research and development. As a specific example, we have launched a joint project with SEC Carbon Co., Ltd. to apply the functional carbon obtained by this method to improving the performance of in-vehicle secondary batteries for electric vehicles (EVs), and are advancing the roadmap toward commercialization and social implementation.
RESEARCH CONTENTSCarbon Plating


I’MSEP Co., Ltd. has achieved "electrolytic carbon plating" by using molten salt as the electrolytic bath—something that was difficult to realize in aqueous systems. This innovative technology is based on electrochemical reactions, just like the widely used electrolytic plating in aqueous systems.
Specifically, using carbide ions (C22–) contained in molten salt as the carbon source and treating a metal substrate as the anode, C22– is oxidized and a carbon plating film is formed on the metal substrate surface. This innovative technology can contribute to improving the performance of energy conversion electrochemical devices to meet the demands of the coming era of electric vehicles (EVs) and 5G. It can also enhance the corrosion resistance of fasteners such as bolts, nuts, and screws. Furthermore, applications to heat transfer and heat dissipation components, electrodes for various electrolysis processes, and biosensors are expected.
This technology is positioned as an important technology for society as it embraces the SDGs and moves toward a carbon-neutral, clean new era.
RESEARCH CONTENTSAmmonia Electrolytic Synthesis

To sustain the lives and livelihoods of a continuously growing human population, increased food production is essential. For this, securing ammonia as a nitrogen fertilizer is necessary. At the same time, hydrogen is expected to play an important role in addressing environmental and energy issues as a storage material, transport medium, and reducing agent for nitrogen oxides. However, current ammonia production relies on the Haber–Bosch process, which has a history of over 100 years.
The current Haber–Bosch process uses natural gas as the hydrogen source, so emissions of carbon dioxide (CO2), a major greenhouse gas, cannot be avoided. Therefore, increasing ammonia production and reducing CO2 have become conflicting challenges. However, the social implementation of the "atmospheric pressure ammonia electrolytic synthesis method" described below can readily resolve this dilemma.
I’MSEP Co., Ltd. has proposed an "atmospheric pressure ammonia electrolytic synthesis method" using molten salt as the electrolytic bath, and is engaged in research and development toward practical application. The following two methods (1) and (2) are presented:
(1) Atmospheric pressure ammonia electrolytic synthesis from hydrogen and nitrogen
Reaction: 1/2 N2 (g) + 3/2 H2 (g) → NH3 (g)
(Theoretical electrolysis voltage 0.05 V)
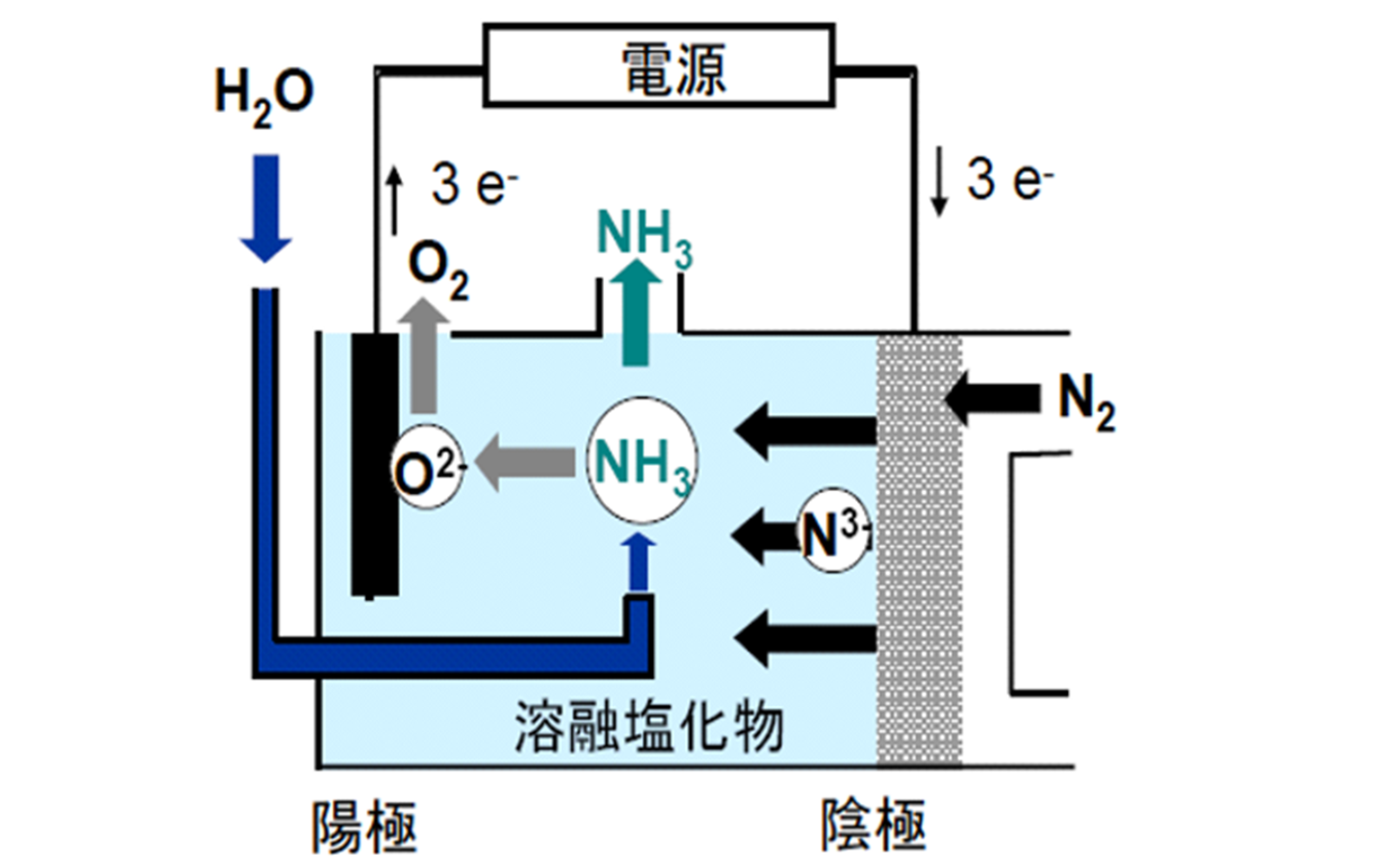
(2) Direct atmospheric pressure ammonia electrolytic synthesis from water and nitrogen
Reaction: 1/2 N2 + 3/2 H2O → NH3 + 3/4 O2
(Theoretical electrolysis voltage 1.17 V)
The feasibility of both methods (1) and (2) has been demonstrated. For example, I’MSEP Co., Ltd. has conducted continuous electrolysis with a prototype electrolysis system cell for method (2) and obtained prospects that continuous electrolysis at a terminal voltage of 2.0 V is possible. Furthermore, we are currently proposing an "advanced electrolysis device" that combines the features of both methods (1) and (2) to enable electrolysis at a terminal voltage of 1.5 V, and are promoting research and development with the goal of its practical application.
RESEARCH CONTENTSRecycling
For example, extracting and recovering only rare earth elements from waste magnets. Elements difficult to separate with conventional technology can be selectively separated and recovered in molten salt.
This enables effective use of a wide range of scarce metals including high-melting-point, high-hardness metals, precious metals, and rare earth metals.
FY2011 Selected for Kyoto City Environmental Future Creation Project
RESEARCH CONTENTSElectrolytic Refining
Molten salt electrolysis for metal refining continues to be used in the production of aluminum, magnesium, rare earth metals, and more. We support the sustainable development of metal refining technology through small-scale electrolysis testing, material development, and electrolyzer structure improvements.
